
The focus of my laboratory is on the molecular mechanisms that play an important role in the pathogenesis and development of different phenotypes of human diabetic retinopathy. By combining whole exome sequencing studies and well harmonized phenotypic analyses of patients and appropriate controls, we are studying a complex disease like diabetic retinopathy. Using cell cultures and animal models, we plan to develop gene regulatory pathways and networks by retinal cell type s, vascular and non-vascular, that will lead to novel therapeutic strategies for treatment of diabetic retinopathy. Our goal is to identify novel genetic variants and proteins for understanding the genetic mechanisms of diabetic retinopathy, and help in development of novel biomarkers, improved therapeutics and precision medicine.
Duration of diabetes is the strongest predictor for progression of diabetic retinopathy (DR), however some diabetics do not develop DR at all, or very mild DR in spite of a long duration of diabetes. Similarly, not all diabetics develop the sight threatening phenotype of diabetic macular edema (DME), or proliferative diabetic retinopathy (PDR). Also, the response to anti-VEGF (vascular endothelial growth factor) drugs in DME is variable with only 30-40% patients (“good responders”) responding well to the treatment. The variability in phenotype of DR and in anti-VEGF treatment responsiveness in DME suggests a potential role for genetic factors. To date, genetic studies – linkage analysis, candidate gene, and genome-wide association (GWAS) – and non-genetic studies of DR have had limited success. Using a well-defined, clinically supported phenotypic strategy, we are currently conducting the DRGen Study to understand better the role of rare genetic variants in DR progression, or protection, and anti-VEGF response in DME. Our collaborators are Harvard’s Joslin Diabetes Center, and Albuquerque’s Indian Health Services (IHS). We are using the whole exome sequencing (WES) to systematically search the entire coding regions of the human genome to detect DR susceptibility genes and variants that cannot be identified through conventional GWAS or linkage analyses.
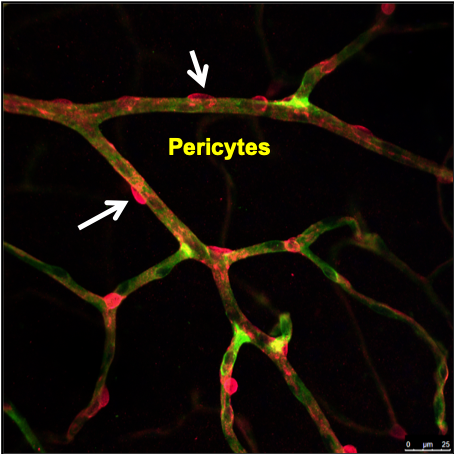
One of the hallmarks of early diabetic retinopathy is the breakdown of the blood-retinal barrier, namely pericyte loss in the human retina. In diabetic experimental animals, pericyte “dropout” appears to include the novel mechanism of pericyte detachment and migration that may be a precursor to the pericyte loss seen in the diabetic retina. The “pericyte dysfunction” in the earliest stages of diabetic retinopathy results in modification of the physical interaction of endothelial cells and pericytes and the ability of pericytes to regulate the endothelial permeability barrier. In addition to the defined roles of PDGF, TGF-β and the angiopoietins in supporting pericyte-endothelial interactions, additional factors may play important roles that could be altered in diseases affecting the retina. Currently, we are examining the transcriptomic profile of isolated pericytes and endothelial cells from diabetic animals. Our RNA transcriptomic work has provided insight into the role of relevant genes in pericytes in the regulation of vascular cell interaction and function in diabetic retinopathy. This work will help us in developing improved therapeutic strategies for sight-threatening diabetic macular edema patients.
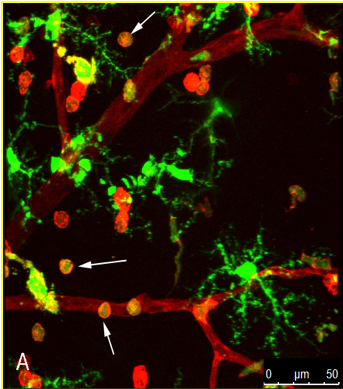
Our recent work explored how inflammation plays a very important role in the alteration of the blood-retinal barrier in diabetic retinopathy. The inflammation cascade sets in with the monocyte trafficking in the retina and microglial activation in diabetes. Chemokines (like CCl2, CCL5) are vital players in this cascade, as their levels are significantly elevated in early diabetic retinopathy. By using knock-out mice and pharmacological approaches, we have established the strategy of chemokine inhibitors in preventing the increased retinal vascular leakage as seen in early diabetes. We further discovered that cathepsin D as a potential mediator in increased retinal endothelial permeability. Also, our team has for the first time emphasized the role of angiopoietin-2 (Ang-2) in alteration of the BRB and its interaction with chemokines. Based on our research work, there are several clinical trials currently going on targeting the Ang-2 pathway in diabetic macular edema and macular degeneration patients.
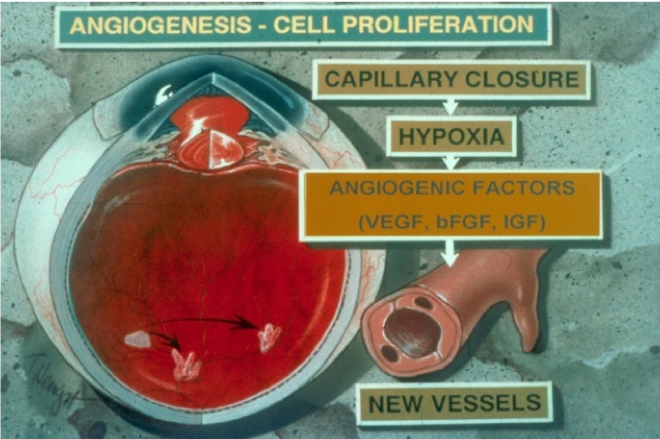
My earlier work addressed the role of proteinases like matrix metalloproteinases (MMPs) and urokinase in retinal and choroidal angiogenesis. We for the first time established the role of these enzymes in the cell migration during the angiogenesis process in retina. We also found novel therapeutic strategies using specific inhibitors of MMPs and urokinase that could successfully suppress retinal angiogenesis in animal models. We further explored the role of the proteinases in alteration of the blood-retinal barrier in diabetic retinopathy. These enzymes were significantly elevated in the retinal tissues in diabetes as shown in the animal models. Our work for the first time showed that these enzymes play a significant role in degrading the cell-cell junctions in retinal vessels in diabetes. This work emphasized the development of proteinase inhibitors in treating retinal angiogenesis and preventing increased retinal vascular permeability and use of these drugs as potential new class of inhibitors in addition to the conventional anti-VEGF agents in diabetic retinopathy.
As a clinical investigator, I have been involved in participating in clinical trials like EDIC (Epidemiology of Diabetes Intervention and Complications), analyzing its results and involved in writing of study results as a member of the Writing Committee of EDIC. This work has emphatically shown the importance of tight blood glucose control in slowing the progression of diabetic retinopathy. Intensive insulin therapy reduces the long-term risk of ocular surgery in patients with type 1 diabetes. We have also participated in many clinical trials with pharmacologic approaches in diabetic retinopathy (READ-2, RIDE/RISE, DRCR Protocol T, BOULEVARD, YOSEMITE).
A Study to Evaluate the Efficacy and Safety of Faricimab (RO6867461) in Participants With Diabetic Macular Edema
Objective: This study will evaluate the efficacy, safety, and pharmacokinetics of faricimab administered at 8-week intervals or as specified in the protocol following treatment initiation, compared with aflibercept once every 8 weeks (Q8W), in participants with diabetic macular edema (DME).
Sponsor: Hoffmann-La Roche
Principal Investigator: Arup Das, MD, PhD
Clinical Coordinator: Marija Zimkute
Long-Term effects of semaglutide on diabetic retinopathy in subjects with type 2 diabetes.
Objective: The objective is to assess the long-term effects of treatment with semaglutide compared to placebo, both added to standard-of-care, on diabetic retinopathy development and progression in subjects with T2D.
Sponsor: Novo Nordisk
Investigator: Arup Das, MD, PhD; Primary Investigator: Mark Burge, MD
Clinical Coordinator: Marija Zimkute
Extension Study for the Port Delivery System With Ranibizumab.
Objective: This study will evaluate the long-term safety and tolerability of the Port Delivery System (PDS) with ranibizumab 100 mg/mL with refills administered every 24 weeks (Q24W) for approximately 144 weeks in participants with neovascular age-related macular degeneration (nAMD) who have completed either Phase II Study GX28228 or Phase III Study GR40548.
Sponsor: Hoffmann-La Roche
Primary Investigator: Bryan Angle, MD
Clinical Coordinator: Marija Zimkute
Diabetes Prevention Program Outcomes Study
Objective: A multi-center clinical trial that examines the efficacy of an intensive lifestyle intervention or metformin to prevent or delay the development of Type 2 diabetes in a population selected to be high-risk due to the presence of impaired glucose tolerance.
Sponsor: National Institutes of Health (NIH)
Investigator: Arup Das, MD, PhD; Primary Investigator: David Schade, MD
Clinical Coordinator: Janene Canady, RN
Epidemiology of Diabetes Interventions and Complications
Objective: This is the national long-term follow-up study of the same group studied in the Diabetes Control and Complications Trial (DCCT). The volunteers in this study have type1 diabetes. EDIC follows these participants to find out if intensive therapy provides risk reduction for retinopathy, nephropathy and neuropathy.
Sponsor: National Institutes of Health (NIH); National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)
Principal Investigator: David Schade, MD
Investigator: Arup Das, MD, PhD
Clinical Coordinator: Janene Canady, RN